Since the industrial revolution, human beings have been promoting industrial development through the massive combustion of fossil fuels. More and more technological demands have made human beings constantly explore renewable resources. Energy from renewable energy should be stored and used when needed, and energy storage and accumulation is the key part of renewable energy utilization.

With the transition from internal combustion engines to electric substitutes, the use of batteries to store energy has been pushed to the forefront. In particular, electrochemical supercapacitor (SC) is more concerned than battery. Its fast storage capacity and enhanced cycle stability have been widely studied in recent years, and it has great potential to fill the gap between battery and existing electrolytic capacitor technology.
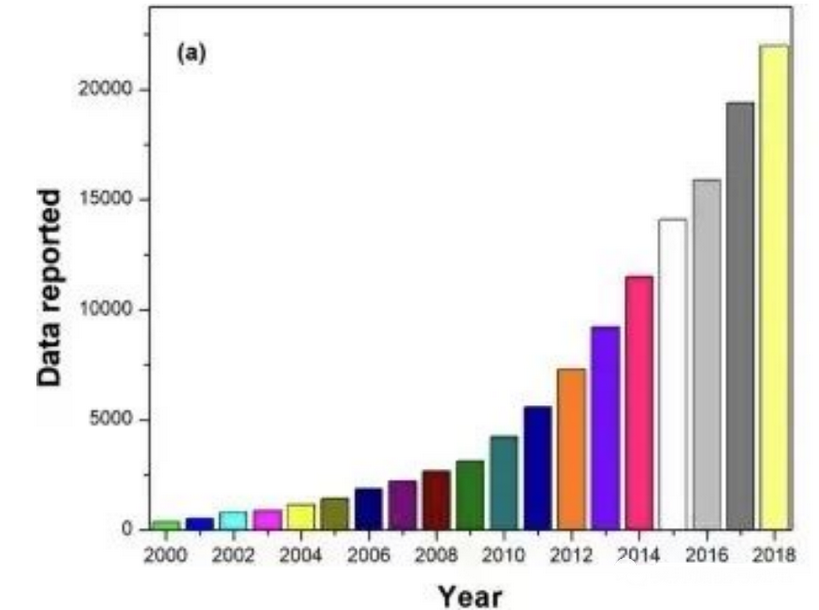
Figure 1 Statistical survey of supercapacitor research activities
What is a supercapacitor
Supercapacitors are also called double-layer capacitors or electrochemical capacitors. The distance between the two charge layers is very small (generally less than 0.5mm). Special electrode structure is used to increase the surface area of the electrode by ten thousand times, resulting in huge capacitance.
Supercapacitor is a new type of energy storage system that has attracted the attention of many scientists in recent years. In short, we can think of supercapacitor as a combination of ordinary capacitor and battery, but it is different from both of them.
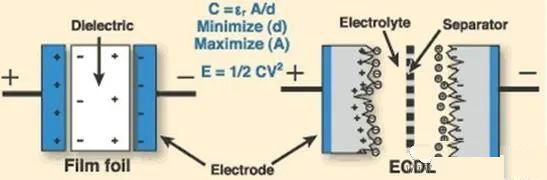
Supercapacitor energy storage application
Supercapacitor energy storage has been developed for more than 50 years. In the past 20 years, technology has made rapid progress, which has greatly increased its capacitance compared with traditional capacitance, reaching the order of thousands of farads, and the specific power density can reach ten times of traditional capacitance.
Supercapacitors store electric energy directly in the electric field, without energy conversion, with fast charging and discharging time, and are suitable for improving power quality. Due to its low energy density, it is suitable for combined use with other energy storage methods.
Types of supercapacitors
Supercapacitors belong to the category of wet electrolytic capacitors. Liquid electrolytes containing ions (charged complexes) are used to ensure charge transfer. According to the principle of energy storage, it can be divided into three categories:
The first category includes double-layer capacitors (EDLC), which use liquid electrolyte non-protonic solvents, such as PC, DEC, DME, etc.
The second category includes the so-called pseudo supercapacitors (Faraday supercapacitors), which are much lower in frequency than EDLC supercapacitors, and are more like a battery in principle.
The third type is hybrid supercapacitors, which combine EDLC and pseudo supercapacitors as the name implies, with higher volume and weight energy density and the ability to provide high current.
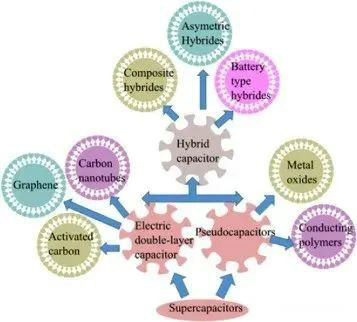
Figure 2 Types of supercapacitors
Difference in working principle between supercapacitors and lithium batteries
Super capacitors and batteries are both electric energy storage systems. If we look at lithium-ion batteries, they are completely dependent on chemical reactions. It is composed of positive electrode and negative electrode, which is technically called anode and cathode.
These two sides are immersed in liquid electrolyte and separated by microporous separator, which only allows ions to pass through. During charging and discharging of lithium battery, ions tend to flow back and forth between anode and cathode.
When these ions are transferred, they will cause the battery to heat, expand, and then shrink. These reactions will gradually degrade the battery, resulting in shorter battery life. However, a significant advantage of lithium battery technology is that it has very high specific energy or energy density and can store energy for future use.
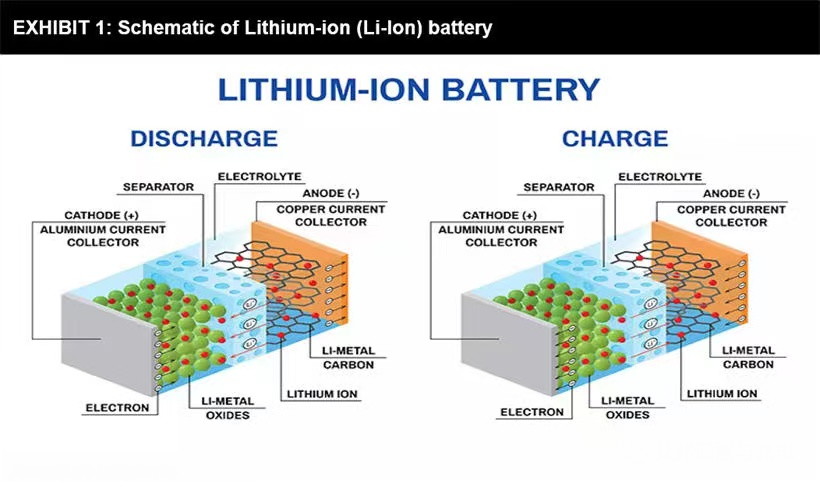
Operating principle of lithium ion battery
But the supercapacitor is different: it does not depend on chemical action to play its role. Instead, it stores electrical energy in an electrostatic manner. The supercapacitor uses a dielectric or insulator between its plates to separate the positive (+ve) and negative (- ve) charges accumulated on each side of the plates. It is this separation that enables the equipment to store energy and release energy quickly.
It can basically capture static electricity for future use. The most obvious advantage of this is that the current 3V capacitor will still be 3V capacitor in 15-20 years. On the contrary, lithium-ion batteries may lose voltage capacity over time and reuse.
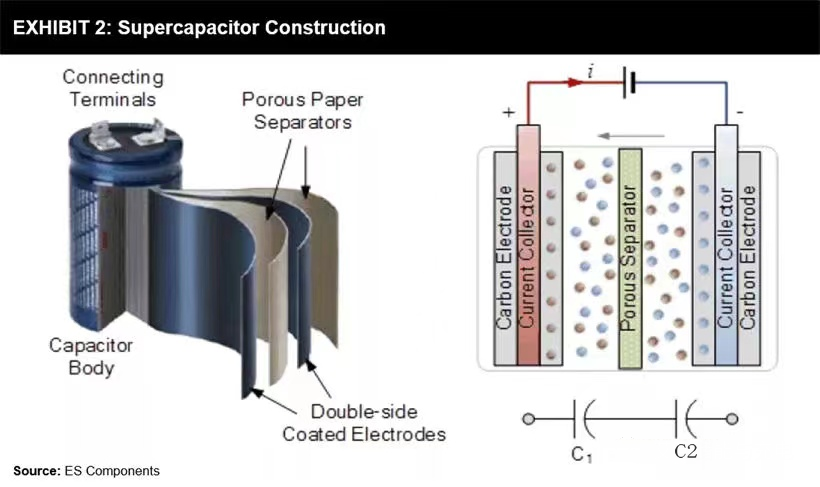
Working principle of supercapacitor
In addition, supercapacitors have higher power throughput than lithium batteries, which means they can be charged and discharged in a very short time. However, compared with battery, its specific energy is very low. Supercapacitors are most suitable for applications with small capacity but power explosion.
What are the advantages of supercapacitors compared with lithium batteries
 Picture of supercapacitor
Picture of supercapacitor
The answer to this question depends largely on its application. Each energy storage technology has some obvious advantages and disadvantages. As mentioned earlier, the energy density of lithium batteries is much higher than that of supercapacitors.
This means that the battery is more suitable for applications with higher energy density, for example, applications where the device needs to run for a long time after a charge. On the other hand, the power density of supercapacitors is much higher than that of batteries. This makes it very suitable for high power consumption applications such as electric vehicle power supply.
The life of supercapacitors is also much longer than that of batteries. Ordinary lithium batteries can handle about 2000-3000 charging and discharging cycles, while supercapacitors can usually maintain more than 1 million charging and discharging cycles, which can save a lot of materials and costs.
Supercapacitors can be charged in a few seconds. Unlike chemical batteries that rely on internal chemical reactions and therefore wear out quickly, supercapacitors will not deteriorate with time. Today's 2.7V supercapacitor will become 2.7V supercapacitor in 15 years. In contrast, all current battery designs will gradually reduce performance, which means that your current 12-volt battery may become 11.4-volt in just three years.
Although supercapacitors with the same weight as lithium-ion batteries can hold more power, their watt/kg (power density) is 10 times higher than lithium-ion batteries. However, the inability of the supercapacitor to discharge slowly means that its watt-hour/kg (energy density) is only a small part of the lithium-ion battery.
Energy storage mechanism
The energy storage mechanism of SC can be explained by three types of capacitive behavior:
(1) Electrochemical double-layer capacitor (EDLC) uses the pure charge accumulated at the electrode interface
(2) Pseudocapacitance (PC) is developed from fast and reversible surface redox process,
(3) The hybrid capacitor utilizes the above two mechanisms
Electrochemical double-layer capacitor
EDLC has the largest effective surface space, the smallest charge separation distance, and higher energy density than traditional capacitors. EDLC was originally a Helmholtz model (the opposite charge was layered at the electrode/electrolyte interface and separated by atomic distance, similar to the traditional two-plate capacitor).
Then, it is modified as the diffusion layer model (at this time, when the point charge is close to the electrode surface, the large capacitance will rise with the increase of the point charge), which is finally interpreted as the combination model of the two.
The conductivity of EDLC on the electrode surface of the planner is controlled by the electric field on the electrode and electrolyte and the chemical affinity between them. Because the electrode has a high specific area of porous structure, the EDLC behavior is highly dependent on porosity.
Therefore, the transport of particles in the confined system is controlled by such factors as the affinity of the tortuous mass transfer path, the restriction of the area in the pores, the electrical phenomena related to the solution and the wettability of the pore surface of the solution.
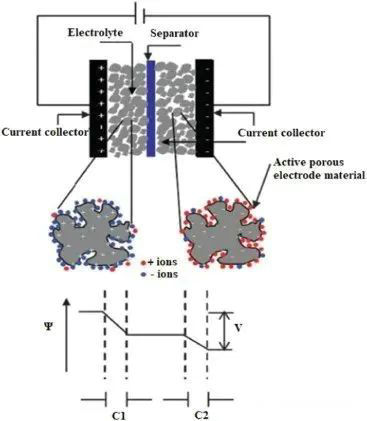
Schematic diagram of EDLC made of porous electrode material
Pseudocapacitance
The PC mechanism is based on Faraday charge process (electron transfer at the electrode interface through oxidation or reduction of chemical substances), which can be reversible or irreversible. In the reversible process, no new chemicals will be produced in the chemical reaction (oxidation/reduction) process, and new chemicals will be produced in the irreversible Faraday process.
The interaction between electrolyte and solid materials involves Faraday reaction to reflect the mechanism of charge transfer, and usually involves surface adsorption, which depends on the surface mechanism.
Advantages and development direction
(1) Supercapacitors can provide hundreds to thousands of times of power in the same volume, which can be applied to applications that need power bursts but do not need high energy storage capacity;
(2) It can not only discharge in a few seconds, but also charge in a very short time, which is a very important advantage for the energy recovery system;
(3) Because of its internal charge storage mechanism, the supercapacitor can withstand millions of cycles and has a cycle life;
(4) In high power rated charging and discharging, supercapacitors are safer than ordinary batteries.
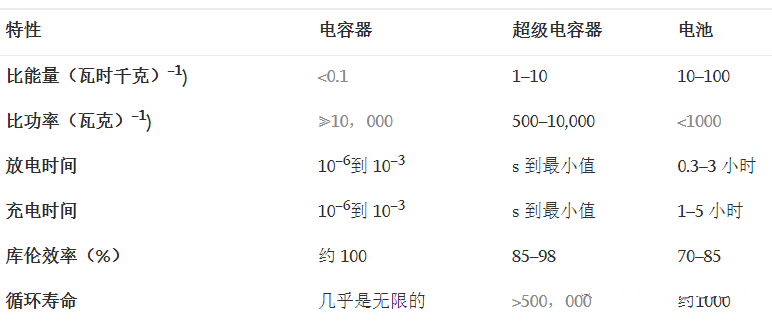
Table 1 Comparison of electrochemical energy storage technology

Table 2 Comparison between battery and supercapacitor













